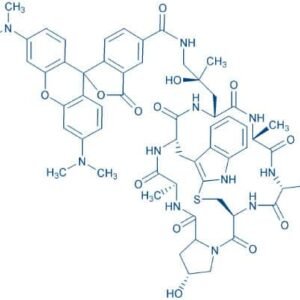
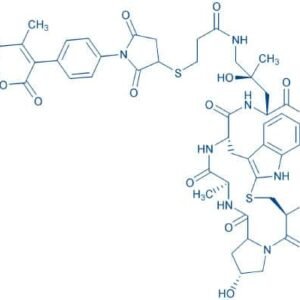
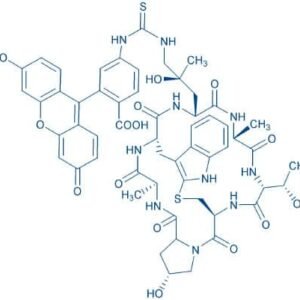
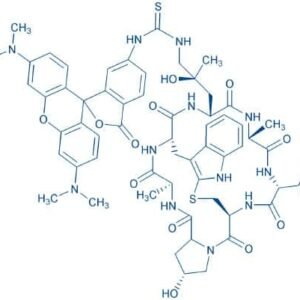
Description
During heparin-induced self-aggregation of the four repeat domain peptides (R1–R4) excised from tau, R1, which resembles Tau Peptide (244-274), was the most resistive to filament formation. Its conformation remained unchanged. The peptide maintained the same random structure under both acidic and neutral conditions.