Description
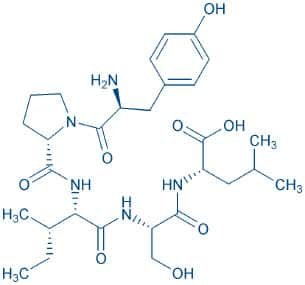
Gluten Exorphin C, isolated from the pepsin-trypsin-chymotrypsin digest of wheat gluten, was considered as a δ-opioid receptor-selective ligand. The hydrophobicity of Ile³ seems to be important for the expression of the opioid activity of gluten exorphin C. Moreover, this peptide appears to be quite different from any of the endogenous and exogenous opioid peptides ever reported as the N-terminal Tyr is the only aromatic amino acid in the structure.